
Bryan Spangelo
Professor Emeritus
Additional Info
My research lab is investigating the anti-cancer potential of novel platinum-containing compounds. This work is performed in collaboration with chemistry professor, Pradip Bhowmik.
Expertise
Immunopharmacology, Neuro-Immune Interactions, Cancer Research, Drug Discovery for Novel Chemotherapeutics
Research Interests
The control of anterior pituitary hormone secretion is subject to multiple levels of regulation. The hypothalamus is connected to the pituitary by a vasculature known as the long portal vessels. Biogenic amines and neuropeptides produced within the hypothalamus traverse these portal vessels to affect pituitary hormone secretion. Target tissues of the anterior pituitary produce hormones that feedback on the hypothalamus and/or pituitary for the regulation of the neuroendocrine axis. The immune system is subject to neuroendocrine control; thus, the products of this system (e.g., interleukins, thymic hormones, cytokines) can regulate anterior pituitary hormone secretion. We have reported that the cytokine interleukin-6 (IL-6) stimulates the release of several anterior pituitary hormones, including prolactin, growth hormone, and luteinizing hormone. Because IL-6 does not normally reach hormone-stimulating concentration in the blood, we hypothesized that this cytokine is produced within the neuroendocrine system.
We have demonstrated the synthesis and release of IL-6 from a cellular subpopulation of the anterior pituitary. The release of IL-6 was enhanced by neuropeptides, IL-1b as well as agents that increased intracellular cAMP. We also reported the production of IL-6 in the medial basal hypothalamus and recently, the neurointermediate pituitary lobe. Thus, all components of the neuroendocrine system are capable of IL-6 production, suggesting a paracrine, facilitative role for this cytokine in hormone secretion.
We have investigated the intracellular signaling pathway activated by IL-1b in the folliculostellate cell, the putative source of IL-6 in the anterior pituitary. Interleukin-1b stimulates the phospholipase A2-mediated hydrolysis of phosphatidylcholine (PtdCho), leading to arachidonic acid liberation and lysophosphatidylcholine (LysoPC) production. The 5-lipoxygenase pathway of arachidonic acid metabolism is necessary for IL-b-mediated IL-6 release. In addition, LysoPC (but not other lyso-lipids) stimulates IL-6 release through the apparent activation of protein kinase C (PKC). Thus, IL-1b may stimulate neuroendocrine IL-6 synthesis and release via a novel pathway. Current work is directed at the complete elucidation of IL-1b activated signal transduction in the anterior pituitary as well as in the rat C6 glioma cell line.
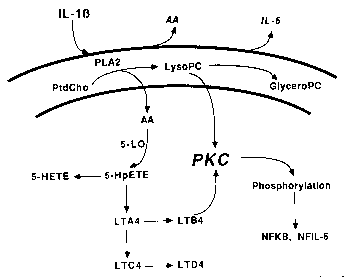
Proposed signal transduction pathway activated by IL-1b resulting in the release of IL-6 from anterior pituitary folliculostellate (FS) cells in vitro. Abbreviations used: IL-1b (interleukin-1b), IL-6 (interleukin-6), AA (arachidonic acid), PtdCho (phosphatidylcholine), PLA2 (phospholipase A2), LysoPC (lysophosphatidylcholine), GlyceroPC (glycerophosphorycholine), 5-LO (5-lipoxygenase), 5-HpETE (5-hydroperoxyeicosatetraenoic acid), 5-HETE (5-hydroxyeicosatetraenoic acid), LTA4 (leukotriene B4), LTC4 (leukotriene C4), LTD4 (leukotriene D4), PKC (protein kinase C), NFKB (nuclear factor kB), NFIL-6 (nuclear factor IL-6).
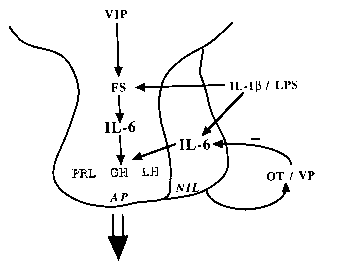
Proposed paracrine model for neuroendocrine IL-6 facilitation of anterior pituitary hormone secretion in vivo. IL-6 production by the folliculo-stellate cells in the anterior pituitary may act in a paracrine manner to affect hormone secretion from the lactotropes, somatotropes, and gonadotropes. In addition, neurointermediate pituitary lobe IL-6 may gain access to the anterior pituitary by the short portal vasculature. Abbreviations used: VIP (vasoactive intestinal polypeptide), FS (folliculo-stellate cells), IL-6 (interleukin-6), PRL (prolactin), GH (growth hormone), LH (luteinizing hormone), AP (anterior pituitary), NIL (neurointermediate pituitary lobe), IL-1b (interleukin-1b), LPS (lipopolysaccharide), OT (oxytocin), VP (vasopressin).
